Prostate Cancer
Prostate cancer is the fourth leading cause of cancer deaths worldwide and the second most common form of cancer and cancer death in men in America. There is a large degree of heterogeneity amongst prostate cancers, both between individual cases and within more specific categories of disease progression, which complicates treatment and further research. Initial diagnosis utilizes immunohistochemistry to determine if a tumor is invasive and if it will respond to androgen therapy.
Jump to product table
Prostate Cancer Product Areas
Localized prostate cancer grows slowly and may not need treatment, or it can be managed with surgery. However, if the cancer grows and metastasizes, primary androgen therapy can be administered to act at androgen receptors and suppress the release of androgens. The same approach can be used in prostate cancer research, for example, Goserelin acetate (Cat. No. 3592) is a synthetic gonadotropin-releasing hormone (GnRH) analog that behaves as an agonist at the GnRH receptors. It stimulates gonadotropin and sex hormone release in the short term and suppresses prostate cancer tumor growth with continued administration. Prostate cancer that is resistant to primary androgen therapy, a state known as metastatic castration resistant prostate cancer (mCRPC), can be treated with second generation androgen deprivation therapy. To simulate this process in research, the enzyme inhibitor Abiraterone acetate (Cat. No. 6520) can be used, which inhibits CYP450 enzymes involved in the synthesis of androgens.
Prostate cancer cells demonstrate many of the same genetic and metabolic changes as other cancer types, such as an increased requirement for nutrients to promote rapid growth. However, there are also some more specific metabolic, enzyme and genetic prostate cancer markers that are the subject of further research. For example, the cells of the most common form of prostate cancer, acinar adenocarcinoma, accumulate large reservoirs of zinc and, unlike other solid tumors, prostate cancer tends to rely more heavily on anaplerosis (TCA cycle) for amino acid metabolism and have elevated TCA cycle intermediates, this compares to a focus solely on energy production from anaplerosis in other cancers.
Other metabolic alterations in prostate cancer include: upregulation of phosophoglycerate dehydrogenase, increased glycolysis due to activation of GLUT1, increased hexokinase 1/2 activity and expression, and the upregulation of G6PD in response to mTOR.
Genetic alterations in prostate cancer include up- and down-regulation of specific genes; for example, AURKA and MYCN are amplified, but there is loss of function of RB1 and TP53/p53 and loss of PTEN. The aurora kinases encoded by AURKA gene are involved in mitosis and MYCN encodes a nuclear protein that drives cell growth and proliferation. RB1 and TP53 are both tumor suppressor genes and PTEN loss leads to hyperactivation of PI3K/AKT/mTOR signaling and is found in almost half of mCRPC tumors.
Prostate Cancer Tumor Progression
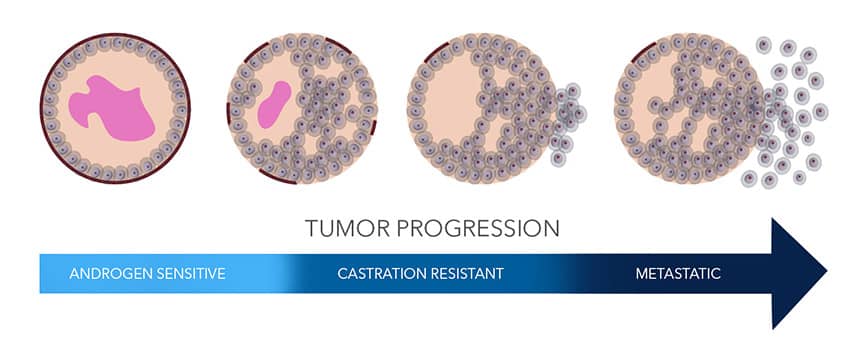
Figure 1: Prostate cancer tumor progression Schematic showing the progression of prostate cancer from normal tissue (left) through to metastatic disease (right).
Prostate Cancer Research Targets
| Prostate cancer marker | Tumor change or target | Tumor progression and targets for research |
|---|---|---|
| Metabolic | Citrate-oriented in normal prostate | Oxidative phosphorylation, lipogenesis (FASN and lipid metabolism), increased glycolysis |
| Hypoxia – angiogenesis and aggressive tumor growth (HIFs) | ||
| Genetic | Loss or loss of function | PTEN loss, loss of function of RB1 and TP53/p53 |
| Upregulation | Aurora kinases; MYC | |
| Epigenetic or transcription factors | Methylation, microRNA, histone modifications | EZH2 overexpression, bromodomains |
| Enzymes | Overexpression | Methyltransferases, hexokinases, G6PD, PI3K, AKT, mTOR, cytochrome P450, matrix metalloproteinases, HDAC1 |
| Androgen receptor | Progressive dysregulation | Receptor downregulation (PROTAC® degraders and targeted protein degradation); Wnt signalling, β-catenin |
| Other targets | RAF, OCT4, BRD4 |
New and Top Products for Prostate Cancer Research
Click product name to view details and order
| Target | Top Products | New Products |
|---|---|---|
| AKT1 | Akti-1/2, SC 79 | AT 7867 |
| AR | Cl-4AS-1, GSK 650394 | ARCC 4 (PROTAC®) |
| AURKA | Lestaurtinib, Anacardic acid | |
| CTNNB1 | endo-IWR 1, XAV 939 (GMP version available) | FzM1.8 |
| CYP17A1 | Abiraterone acetate | |
| ERG | ERGi-USU | |
| EZH2 | 3-Deazaneplanocin A hydrochloride, UNC 1999 | JQEZ5 |
| NR3C1 | Gap 27, Hydrocortisone | Alsterpaullone |
| GSK-3B | CHIR 99021, SB 216763 | |
| HSPA5 | Pifithrin-μ, VER 155008 | |
| MYC | 10058-F4, KJ Pyr 9 | |
| NCOA1 | Lys-CoA | |
| PIK3CA/PIK3CB | LY 294002 hydrochloride, Wortmannin | Omipalisib |
| TMPRSS2 | Nafamostat mesylate | |
| FOXM1 | RCM 1 | |
| BRD4 | MZ 1, (+)-JQ1 | AT 1 (PROTAC®) |
| CUL3 | WS 383 | |
| BMI1 | PRT 4165 | |
| Asah1 | Oleylethanolamide | |
| FOXA1 | (+)-JQ1 | |
| ABCG2 | Kyoto Probe-1 | |
| NR2F2 | CIA 1 | |
| SPOP | SPOP-i-6lc |
PROTAC® is a registered trademark of Arvinas Operations, Inc., and is used under license.
Literature for Prostate Cancer
Tocris offers the following scientific literature for Prostate Cancer to showcase our products. We invite you to request* or download your copy today!
*Please note that Tocris will only send literature to established scientific business / institute addresses.
Cell Cycle and DNA Damage Research Product Guide
This product guide provides a review of the cell cycle and DNA damage research area and lists over 150 products, including research tools for:
- Cell Cycle and Mitosis
- DNA Damage Repair
- Targeted Protein Degradation
- Ubiquitin Proteasome Pathway
- Chemotherapy Targets
TPD and Induced Proximity Research Product Guide
This brochure highlights the tools and services available from Bio-Techne to support your Targeted Protein Degradation and Induced Proximity research, including:
- Active Degraders
- TAG Degradation Platform
- Degrader Building Blocks
- Assays for Protein Degradation
- Induced Proximity Tools
MAPK Signaling Scientific Review
MAP kinase signaling is integral to the regulation of numerous cellular processes such as proliferation and differentiation, and as a result is an important focus of cancer and immunology research. Updated for 2016, this review discusses the regulation of the MAPK pathway and properties of MAPK cascades. Compounds available from Tocris are listed.
Cancer Metabolism Poster
This poster summarizes the main metabolic pathways in cancer cells and highlights potential targets for cancer therapeutics. Genetic changes and epigenetic modifications in cancer cells alter the regulation of cellular metabolic pathways providing potential cancer therapeutic targets.
Epigenetics in Cancer Poster
This poster summarizes the main epigenetic targets in cancer. The dysregulation of epigenetic modifications has been shown to result in oncogenesis and cancer progression. Unlike genetic mutations, epigenetic alterations are considered to be reversible and thus make promising therapeutic targets.
Programmed Cell Death Poster
There are two currently recognized forms of programmed cell death: apoptosis and necroptosis. This poster summarizes the signaling pathways involved in apoptosis, necroptosis and cell survival following death receptor activation, and highlights the influence of the molecular switch, cFLIP, on cell fate.










