Submit a Review & Earn an Amazon Gift Card
You can now submit reviews for your favorite Tocris products. Your review will help other researchers decide on the best products for their research. Why not submit a review today?!
Submit ReviewDoxazosin mesylate is a selective α1-adrenoceptor antagonist (pKi values are 9.0, 8.5 and 8.4 for human α1B, α1A and α1D receptors respectively). Displays antihypertensive activity.
 View Larger
View Larger
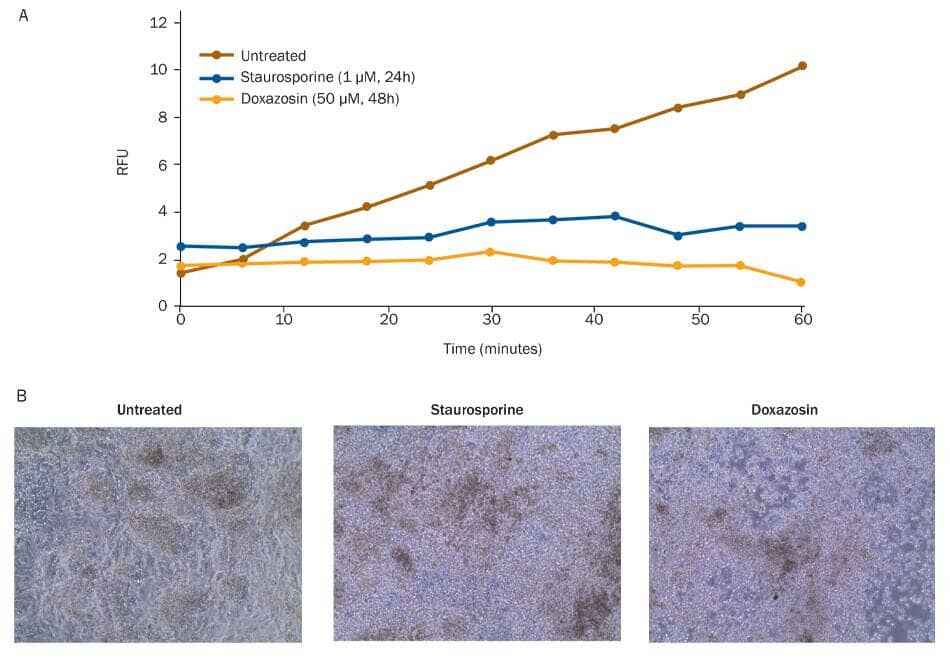
Reduced viability of differentiated cardiomyocytes exposed to cardiotoxic small molecules. The viability of differentiated cardiomyocytes was assessed using a Resazurin metabolism assay. A) Untreated cells metabolize Resazurin (Catalog # AR002) to produce resorufin, a fluorescent molecule that can be measured using a fluorometric plate reader. Resorufin fluorescence accumulated in untreated cardiomyocytes. Cells treated with the cardiotoxic small molecules Staurosporine (blue; Catalog # 1285), a non-selective protein kinase inhibitor, or Doxazosin (tan; Catalog # 2964), an alpha 1 antagonist, did not metabolize Resazurin, as shown by lack of fluorescence accumulation, indicating a loss of viability upon treatment. B) Cell morphology of untreated, Staurosporine-treated, and Doxazosin-treated cardiomyocytes was assessed by brightfield microscopy.
Doxazosin mesylate is also offered as part of the Tocriscreen 2.0 Max and Tocriscreen FDA-Approved Drugs. Find out more about compound libraries available from Tocris.
| M. Wt | 547.58 |
| Formula | C23H25N5O5.CH3SO3H |
| Storage | Desiccate at RT |
| Purity | ≥98% (HPLC) |
| CAS Number | 77883-43-3 |
| PubChem ID | 62978 |
| InChI Key | VJECBOKJABCYMF-UHFFFAOYSA-N |
| Smiles | NC2=NC(N3CCN(C(C4COC(C=CC=C5)=C5O4)=O)CC3)=NC1=CC(OC)=C(OC)C=C12.CS(=O)(O)=O |
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Tocris products are intended for laboratory research use only, unless stated otherwise.
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 41.07 | 75 |
The following data is based on the product molecular weight 547.58. Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which will affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.75 mM | 2.43 mL | 12.17 mL | 24.35 mL |
| 3.75 mM | 0.49 mL | 2.43 mL | 4.87 mL |
| 7.5 mM | 0.24 mL | 1.22 mL | 2.43 mL |
| 37.5 mM | 0.05 mL | 0.24 mL | 0.49 mL |
References are publications that support the biological activity of the product.
Alabaster and Davey (1986) The α1-adrenoceptor antagonist profile of doxazosin: preclinical pharmacology. Br.J.Clin.Pharmacol. 21 9S PMID: 2871857
Kenny et al (1996) Evaluation of the pharmacological selectivity profile of α1 adrenoceptor antagonists at prostatic α1 adrenoceptors: binding functional and in vivo studies. Br.J.Pharmacol. 118 871 PMID: 8799556
Ma et al (2006) Chiral selective effects of doxa. enantiomers on blood pressure and urinaruy bladder pressure in anesthetized rats. Acta Pharmacol.Sinica 27 1423 PMID: 17049117
If you know of a relevant reference for Doxazosin mesylate, please let us know.
Keywords: Doxazosin mesylate, Doxazosin mesylate supplier, α1-adrenoceptor, alpha1-adrenoceptor, a1-adrenergic, α1-adrenergic, alpha1-adrenergic, a1-adrenoceptor, antagonists, Receptors, UK33274, UK, 33274, Adrenergic, Alpha-1, 2964, Tocris Bioscience
Citations are publications that use Tocris products. Selected citations for Doxazosin mesylate include:
Diaz et al (2014) Moderate Alcohol Exposure during the Rat Equivalent to the Third Trimester of Human Pregnancy Alters Regulation of GABAA Receptor-Mediated Synaptic Transmission by DA in the Basolateral Amygdala. Front Cell Neurosci 2 46 PMID: 24904907
Jahchan et al (2013) A drug repositioning approach identifies tricyclic antidepressants as inhibitors of small cell lung cancer and other neuroendocrine tumors. Cancer Discov 3 1364 PMID: 24078773
Kahn et al (2016) The anti-hypertensive drug pra. inhibits glioblastoma growth via the PKCδ-dependent inhibition of the AKT pathway. EMBO Mol Med 8 511 PMID: 27138566
Cilz et al (2014) DArgic modulation of GABAergic transmission in the entorhinal cortex: concerted roles of α1 adrenoreceptors, inward rectifier K+, and T-type Ca2+ channels. Cereb Cortex 24 3195 PMID: 23843440
Do you know of a great paper that uses Doxazosin mesylate from Tocris? Please let us know.
Average Rating: 4 (Based on 1 Review.)
$25/€18/£15/$25CAN/¥75 Yuan/¥2500 Yen for a review with an image
$10/€7/£6/$10 CAD/¥70 Yuan/¥1110 Yen for a review without an image
Filter by:
The drug was incubated at 10 μM to investigate a potential effect on cell metabolism.
Tocris offers the following scientific literature in this area to showcase our products. We invite you to request* your copy today!
*Please note that Tocris will only send literature to established scientific business / institute addresses.
Major depressive disorder is characterized by depressed mood and a loss of interest and/or pleasure. Updated in 2015 this poster highlights presynaptic and postsynaptic targets for the potential treatment of major depressive disorder, as well as outlining the pharmacology of currently approved antidepressant drugs.