Submit a Review & Earn an Amazon Gift Card
You can now submit reviews for your favorite Tocris products. Your review will help other researchers decide on the best products for their research. Why not submit a review today?!
Submit ReviewPropranolol hydrochloride is an β-adrenergic antagonist.
R-enantiomer, S-enantiomer and Glycol Metabolite also available.
 View Larger
View Larger
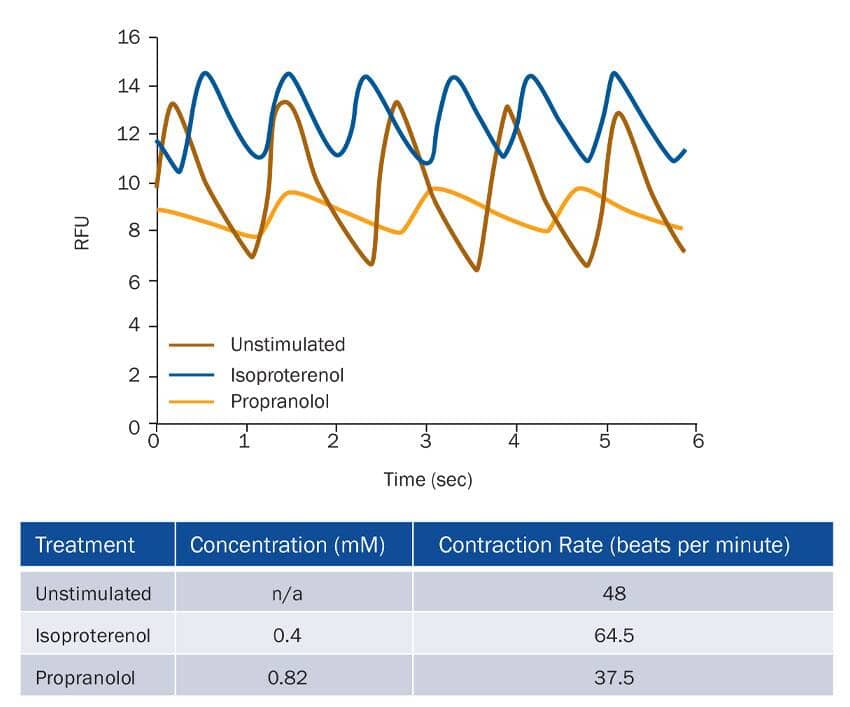
Small molecules affect the rate of cardiomyocyte contraction. Cardiomyocytes were differentiated from the BG01V human embryonic stem cells and assessed for their ability to contract using the Fluo-4 calcium binding assay. Rate interval graph show fluctuations in Fluo-4 fluorescence intensity, indicative of cardiomyocyte contraction, under basal conditions (brown; untreated) and following sequential treatment with Isoproterenol (blue; Catalog # 1747), a beta -adrenergic agonist, and Propranolol (tan; Catalog # 0624), a beta -adrenergic antagonist. Table shows drug concentrations and contraction rate of cardiomyocytes during each treatment. The contraction rate of differentiated cardiomyocytes was determined as the number of contractions (contraction-interval plus relaxation interval) per minute.
| M. Wt | 295.81 |
| Formula | C16H21NO2.HCl |
| Storage | Store at RT |
| CAS Number | 318-98-9 |
| PubChem ID | 62882 |
| InChI Key | ZMRUPTIKESYGQW-UHFFFAOYSA-N |
| Smiles | Cl.CC(C)NCC(O)COC1=C2C=CC=CC2=CC=C1 |
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Tocris products are intended for laboratory research use only, unless stated otherwise.
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 29.58 | 100 |
The following data is based on the product molecular weight 295.81. Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which will affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 1 mM | 3.38 mL | 16.9 mL | 33.81 mL |
| 5 mM | 0.68 mL | 3.38 mL | 6.76 mL |
| 10 mM | 0.34 mL | 1.69 mL | 3.38 mL |
| 50 mM | 0.07 mL | 0.34 mL | 0.68 mL |
References are publications that support the biological activity of the product.
Litwin et al (1999) Effects of propra. treatment on left ventricular function and intracellular calcium regulation in rats with postinfarction heart failure. Br.J.Pharmacol. 127 1671 PMID: 10455325
Xie et al (1998) The inhibitory effect of propra. on ATP-sensitive potassium channels in neonatal rat heart. Br.J.Pharmacol. 123 599 PMID: 9517376
Merck Index 12 8025
If you know of a relevant reference for Propranolol hydrochloride, please let us know.
Keywords: Propranolol hydrochloride, Propranolol hydrochloride supplier, β-adrenoceptor, beta-adrenoceptors, b-adrenoceptors, b-adrenergic, antagonists, β-adrenergic, beta-adrenergic, Receptors, Non-Selective, β-blockers, beta-blockers, 3506-09-0, Non-selective, Adrenergic, Beta, 0624, Tocris Bioscience
Citations are publications that use Tocris products. Selected citations for Propranolol hydrochloride include:
Gherbi et al (2015) Negative cooperativity across β1-adrenoceptor homodimers provides insights into the nature of the secondary low-affinity CGP 12177 β1-adrenoceptor binding conformation. FASEB J 29 2859 PMID: 25837585
Stoddart et al (2015) Application of BRET to monitor ligand binding to GPCRs. Nat Methods 12 661 PMID: 26030448
Meitzen et al (2011) β1-Adrenergic receptors activate two distinct signaling pathways in striatal neurons. J Neurochem 116 984 PMID: 21143600
Rankovic et al (2011) Modulation of calcium-dependent inactivation of L-type Ca2+ channels via β-adrenergic signaling in thalamocortical relay neurons. PLoS One 6 e27474 PMID: 22164209
McKeithan et al (2017) An Automated Platform for Assessment of Congenital and Drug-Induced Arrhythmia with hiPSC-Derived Cardiomyocytes. Front Physiol 8 766 PMID: 29075196
Bucher et al (2014) Medullary NE neurons modulate local oxygen concentrations in the bed nucleus of the stria terminalis. J Cereb Blood Flow Metab 34 1128 PMID: 24714037
Gherbi et al (2014) Detection of the secondary, low-affinity β1 -adrenoceptor site in living cells using the fluorescent CGP 12177 derivative BODIPY-TMR-CGP. Br J Pharmacol 171 5431 PMID: 25052258
Beas et al (2020) A ventrolateral medulla-midline thalamic circuit for hypoglycemic feeding Nat Commun 11 6218 PMID: 33277492
Kilpatrick et al (2019) Complex Formation between VEGFR2 and the β2-Adrenoceptor. Cell Chem Biol 26 830 PMID: 30956148
Hansen and Manahan-Vaughan (2015) Locus Coeruleus Stimulation Facilitates Long-Term Depression in the Dentate Gyrus That Requires Activation of β-Adrenergic Receptors. Mol Cancer 25 1889 PMID: 24464942
Patsouris et al (2015) Burn Induces Browning of the Subcutaneous White Adipose Tissue in Mice and Humans. Cereb Cortex 13 1538 PMID: 26586436
Skelly and Weiner (2014) Chronic treatment with pra. or dulox. lessens concurrent anxiety-like behavior and alcohol intake: evidence of disrupted noradrenergic signaling in anxiety-related alcohol use. Brain Behav 4 468 PMID: 25161814
Shi et al (2010) Catecholamine up-regulates MMP-7 expression by activating AP-1 and STAT3 in gastric cancer. Purinergic Signal 9 269 PMID: 20939893
Do you know of a great paper that uses Propranolol hydrochloride from Tocris? Please let us know.
Average Rating: 5 (Based on 1 Review.)
$25/€18/£15/$25CAN/¥75 Yuan/¥2500 Yen for a review with an image
$10/€7/£6/$10 CAD/¥70 Yuan/¥1110 Yen for a review without an image
Filter by:
Tocris offers the following scientific literature in this area to showcase our products. We invite you to request* your copy today!
*Please note that Tocris will only send literature to established scientific business / institute addresses.
Major depressive disorder is characterized by depressed mood and a loss of interest and/or pleasure. Updated in 2015 this poster highlights presynaptic and postsynaptic targets for the potential treatment of major depressive disorder, as well as outlining the pharmacology of currently approved antidepressant drugs.