Protocol for LipoLIVE™ Green Non-Toxic Dye
This is intended as a guide only.
In Brief Download the PDF of this protocol
LipoLIVE™ Green Non-Toxic Dye is a ready-to-use fluorescent probe for the real-time visualization of lipid droplet and neutral lipids in live cells. It does not require cell fixation or affect cell health and maintains the physiological integrity of models, enabling free-wash monitoring of lipid synthesis, storage, and mobilization.
LipoLIVE™ is a trademark of Saguaro Biosciences.
1. Protocol Overview
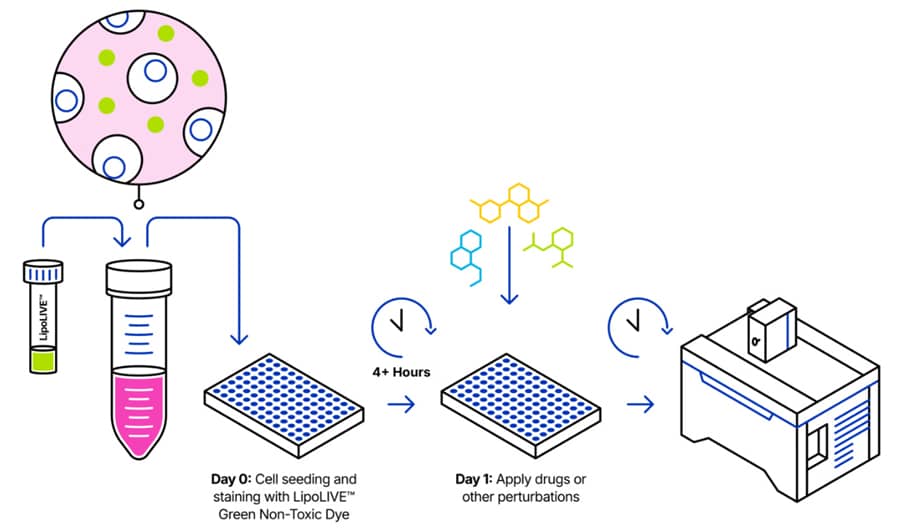
2. Content and Storage
| Product | Content | Storage | Stability |
|---|---|---|---|
|
LipoLIVE™ Green Non-Toxic Dye |
Diluted in 50 µl of DMSO |
-20º C Delivered at room temperature Protect from light |
1 year |
Table 1. LipoLIVE™ Green Non-Toxic Dye Product Information
Intended Use: For research use only. Not for use in diagnostics or therapeutic procedures.
3. General Guidelines
LipoLIVE™ Green Non-Toxic Dye dilution and preparation
- Warm up the LipoLIVE™ Green Non-Toxic Dye tube to room temperature before use to avoid condensation to form and water to get into the anhydrous dye solution.
- Gently spin the tube before use to collect any dye solution that may remain near the cap.
- Dilute LipoLIVE™ Green Non-Toxic Dye 1,000-fold in preferred culture medium.
- Vortex thoroughly.
NOTE: We recommend 1X as a starting point for optimization. Higher or lower concentrations may be optimal for different imaging systems and cell models.
- Seed cells at desired density (typically to achieve 70-80% confluence) in cell culture medium containing LipoLIVE™ Green Non-Toxic Dye in a black multi-well plate. Return to the incubator at 37°C, 5% CO2 overnight.
- No washing step is required prior to imaging. Keep LipoLIVE™ Green Non-Toxic Dye in solution throughout the assay.
Alternative Cell Culture Indications for LipoLIVE™ Green Non-Toxic Dye
- While we recommend seeding cells in the presence of diluted LipoLIVE™ Green Non-Toxic Dye, the dye can be added after cell seeding, before or following compound addition. Optimisation of seeding density and incubation prior to imaging are required. For reference, LipoLIVE™ Green Non-Toxic Dye staining stabilizes after 4 hours in U2OS cells (at a concentration of 1X).
- A nuclear dye can be added to allow cell segmentation during data analysis, such as NucleoLIVE™ Non-Toxic Dye (Cat. No. 8935) or NucleoLIVE™ Blue Non-Toxic Dye (Cat. No. 9011). We recommend running a preliminary imaging test on cells treated with single dyes to validate the staining kinetics and absence of fluorescence bleed-through between the nuclear dye and LipoLIVE™ Green channels on your system.
4. Technical Specifications & Instrument compatibility
LipoLIVE™ Green Non-Toxic Dye is a green fluorescent dye that is compatible with GFP, Cy3 or FITC filters.
Instrument compatibility of LipoLIVE™ Green Non-Toxic Dye
| Manufacturer | Instrument | Filters | Mode |
|---|---|---|---|
| Molecular Devices | ImageXpress Confocal ImageXpress Confocal HT.ai |
Cyan / FITC / Cy3 | Widefield / Confocal |
| PerkinElmer / Revvity | Opera Phenix Opera Phenix Plus Operetta CLS |
488 / 500-550 |
Widefield only Widefield / Confocal Widefield / Confocal |
| Yokogawa | CQ1 | 488 / 525/50 | Confocal |
| CV8000 | 488 / 525/50 | Confocal |
LipoLIVE™ Green Non-Toxic Dye is compatible with other high-content imagers and confocal microscopes.
5. LipoLIVE™ Green Non-Toxic Dye, allowing for high quality staining of lipid droplets and neutral lipids without compromising cell health
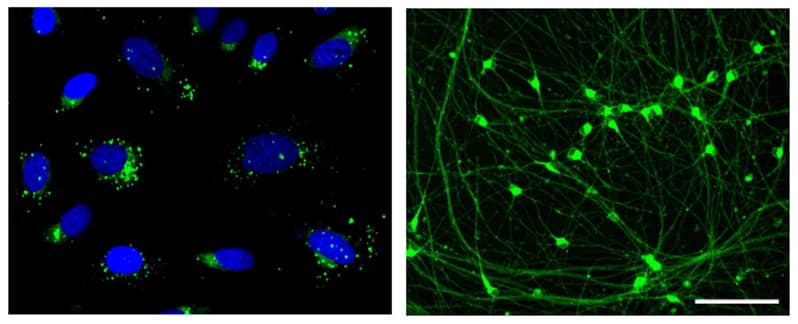
Figure 2. U2OS cells (left) and iPSC-derived neurons (right) stained with LipoLIVE™ Green Non-Toxic Dye. Composite images of cells stained LipoLIVE™ Green Non-Toxic Dye for 72h.
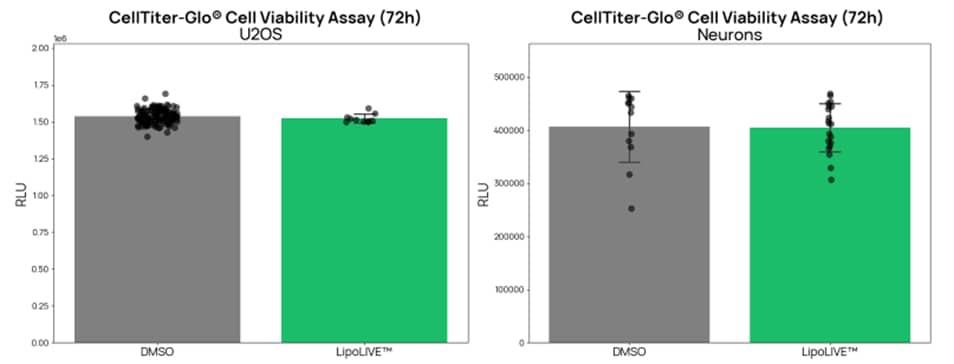
Figure 3. LIPOLIVE™ Green Non-Toxic Dye preserves normal cell proliferation and viability, even in sensitive models. U2OS cells and iPSC-derived neurons showed comparable viability to DMSO controls after 72-hour incubation with LIPOLIVE™ Green Non-Toxic Dye.
6. Recommended Positive Control Compounds for Lipid Studies*
Table 2. Compounds and concentration for U2OS cells in 2D.
| Compound | Mechanism | Concentrations |
|---|---|---|
|
Oleic Acid |
Long-chain monounsaturated fatty, stimulating the rapid formation of cytosolic lipid droplets. | 100 nM-10 μM |
| Palmitic Acid | Long-chain saturated fatty acid, inducing lipotoxic stress and the formation of small clustered cytosolic lipid droplets. | 5 μM-500 μM |
* Compounds provided as examples only. Validation required for each experimental protocol.
7. Example Protocol (for kinetic, 2D live-cell assay)
U2OS cells are treated with standard compounds for ER stress and apoptosis. MCF7 are cultured in DMEM/F-12 complemented with 10% FBS and 1% Penicillin/Streptomycin.
LipoLIVE™ Green Non-Toxic Dye Dilution and Preparation (Day 0):
- Warm up the LipoLIVE™ Green Non-Toxic Dye tubes to room temperature before use and gently spin to collect any dye solution that may remain near the cap.
- Optional: Dilute 10 μL NucleoLIVE™ Non-Toxic Dye in the same 10 mL culture medium (1000-fold).
- Vortex thoroughly.
Cell Culture Protocol with LipoLIVE™ Green Non-toxic Dye (Day 0):
- Harvest and count MCF7 cells.
- Resuspend cells in prepared culture medium with LipoLIVE™ Green Non-Toxic Dye (and NucleoLIVE™ Non-Toxic Dye, if applicable) at 80,000 cells/mL.
- Seed 96-well plate with 100 μL cell suspension per well to a final density of 8,000 cells per well.
- Incubate overnight at 37°C, 5% CO2.
Standard Compound Preparation and Addition (Day 1):
- Prepare dose-response curves with 10x concentrations, maintaining constant vehicle (0.1% DMSO) solvent concentration.
- Prepare negative controls with 0.1% DMSO in complete media.
- Distribute 12.5 μL of test compounds or controls per well.
Imaging and Data Acquisition (Days 1-3):
- Image 96-well plate at 3h, 6h and 24h after addition of test compounds