Protocol for Liperfluo
This is intended as a guide only.
In Brief Download the PDF of this protocol
Liperfluo is used for lipid peroxide detection, and it emits intense fluorescence by the lipid peroxide specific oxidation in organic solvents such as ethanol. Since the excitation and emission wavelengths of the oxidized form of Liperfluo are 524 nm and 535 nm, respectively, both photo-damage against a sample and an auto-fluorescence from the sample can be minimized. Due to the introduction of a tetraethyleneglycol group at the one-end of diisoquinoline ring, the dispersibility of the molecule in an aqueous buffer is improved. Liperfluo oxidized form is almost non-fluorescent in an aqueous media, it emits fluorescence in lipophilic sites such as in cell membranes. Therefore, it can be easily applied for a lipid peroxide imaging by fluorescence microscopy or lipid peroxide analysis by flow cytometry for a living cell.
1. Content and Storage
| Product | Content | Storage |
|---|---|---|
| Liperfluo | 50 µg x 5 |
0-5 °C Protect from light |
Table 1. Liperfluo Product Information
2. Preparation of Solutions
Preparation of a 1 mmol/l Liperfluo DMSO solution
• Add 60 μl of DMSO to a tube containing Liperfluo
• Protect the tube from light with alumininum foil and vortex it for about 2 minutes
Once prepared, Liperfluo DMSO solution should be protected from light and used within 1 day
Preparation of a Liperfluo Working solution
The concentration of Liperfluo for optimal staining will vary depending on the experimental conditions:
• For microscopic imaging: Dilute the 1 mmol/l Liperfluo DMSO solution with serum-free culture medium to prepare Liperfluo working solution
• For flow cytometry: Add the 1 mmol/l Liperfluo DMSO solution directly to cell suspension to be stained
3. General Protocol
Detection of lipid peroxide in the cells
• Seed the cells in a dish or plate
• Remove the supernatant and wash the cells with serum-free culture medium once
• Add an appropriate volume of Liperfluo working solution and incubate at 37°C for 30 minutes
*Optimize the final concentration of Liperfluo because its optimal concentration depends on experimental conditions and other factors
• Remove the supernatant and wash the cells with serum-free culture medium twice
• Observe the cells under a fluorescence microscope or analyze the cells using a flow cytometer
4. Experimental Example
Detection of lipid peroxide by flow cytometry
• HeLa cells in MEM (containing 10% fetal bovine serum) were seeded (3.0×104 cells/well) in a 8 well slide (ibidi) and cultured overnight at 37°C in a 5% CO2 incubator
• Removed the supernatant and the cells were washed with serum-free culture medium once
• Serum-free culture medium (200 μl) containing 1 μmol/l Liperfluo was added to well and the cells were incubated at 37°C for 30 minutes in a 5% CO2 incubator
• The medium was removed, and the cells were washed with 200 μl of HBSS twice
• HBSS (200 μl) containing 500 μmol/l t-BHP (tert-buthyl hydroperoxide) was added to the well and the cells were incubated for 60 minutes at 37°C in a 5% CO2 incubator
• The cells were observed under a confocal fluorescence microscope. (Excitation: 488 nm, Emission: 500-550 nm)
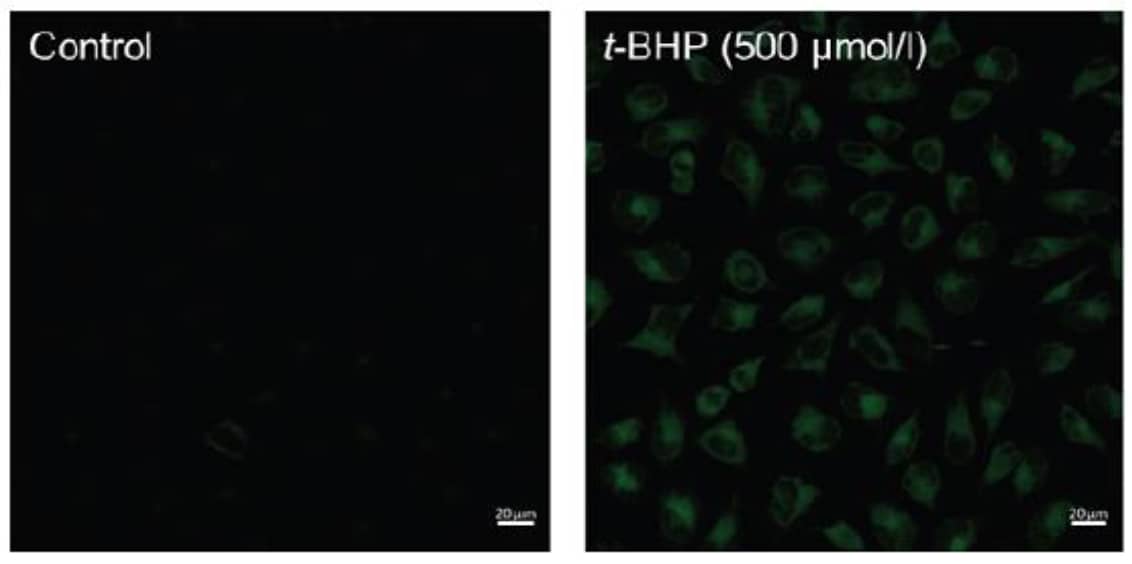
Figure 1: Fluorescence images of lipid peroxidation in HeLa cells
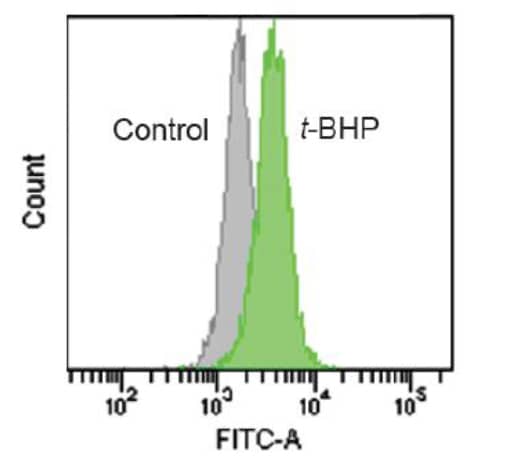
Detection of lipid peroxide by flow cytometry
• HeLa cells in MEM (containing 10% fetal bovine serum) were seeded (1.0×105 cells/well) in a 6 well plate and cultured overnight at 37°C in a 5% CO2 incubator
• Removed the supernatant and the cells were washed with serum-free culture medium once
• Serum-free culture medium (2 ml) containing 1 μmol/l Liperfluo was added to well and the cells were incubated at 37°C for 30 minutes in a 5% CO2 incubator
• The medium was removed, and the cells were washed with 2 ml of HBSS twice
• HBSS (2 ml) containing 500 μmol/l t-BHP (tert-buthyl hydroperoxide) was added to the well, and the cells were incubated for 60 minutes at 37°C in a 5% CO2 incubator
• The cells were harvested by trypsinization and collected into a microtube with culture medium
• The supernatant was replaced with HBSS, and the cells were analyzed using a flow cytometer (Excitation: 488 nm, Emission: 515-545 nm)
Detection of lipid peroxide induced by ferroptosis with erastin (inhibitor for cystine transporter)
• A549 cells in DMEM (containing 10% fetal bovine serum) were seeded (1.0×105 cells/well) in a 8 well slide (ibidi) and cultured overnight at 37°C in a 5% CO2 incubator
• The DMEM was removed and 200 μl of serum-free culture medium containing 50 μmol/l erastin was added to well
• The cells were incubated at 37°C overnight in a 5% CO2 incubator
• The medium was removed, and the cells were washed with 200 μl of HBSS twice
• HBSS (200 μl) containing 5 μmol/l Liperfluo was added to well and the cells were incubated at 37°C for 30 minutes in a 5% CO2 incubator
• The supernatant was removed, and the cells were washed with 200 μl of HBSS twice
• The cells were observed under a confocal fluorescence microscope. (Excitation: 488 nm, Emission: 500-550 nm)